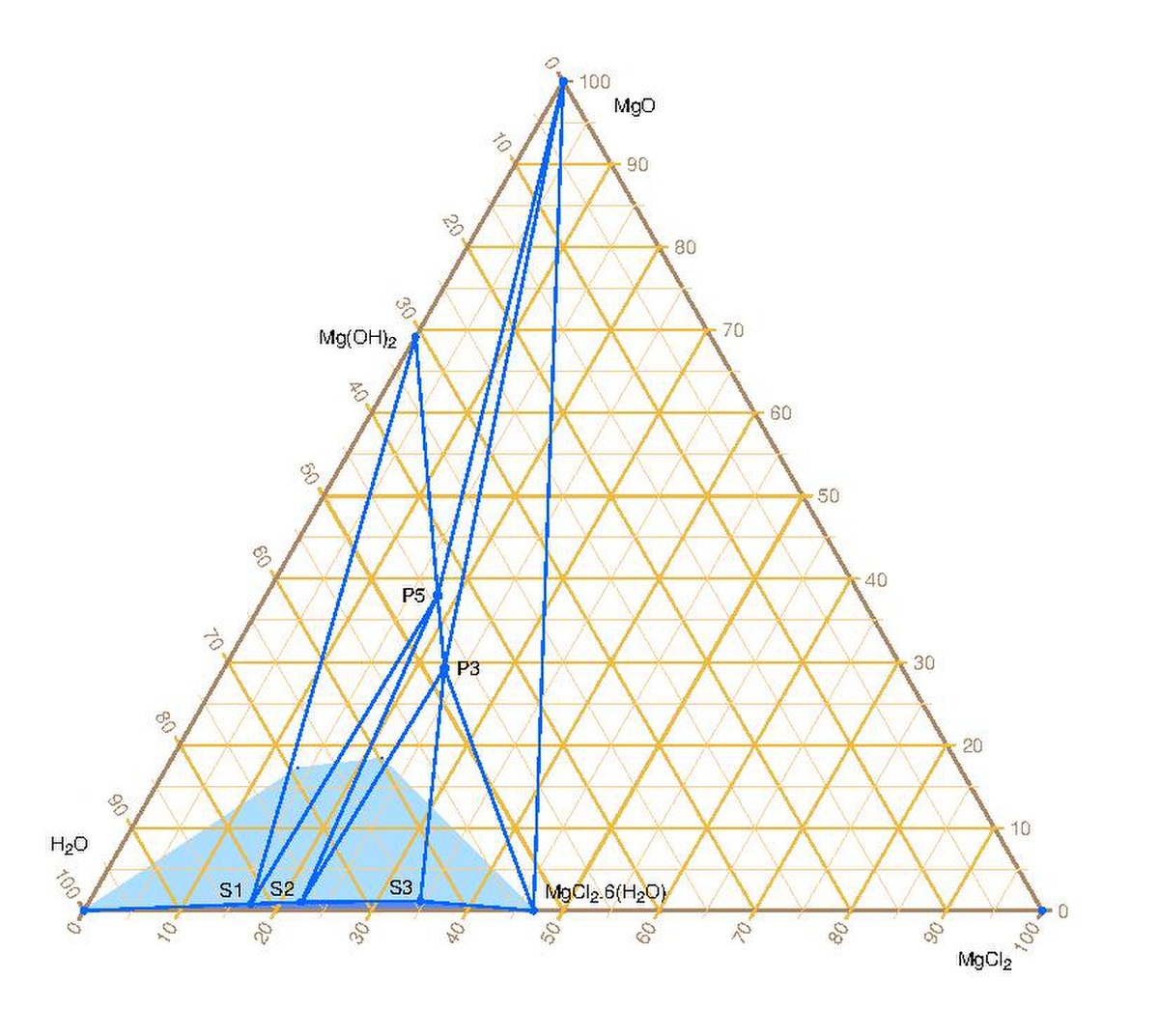
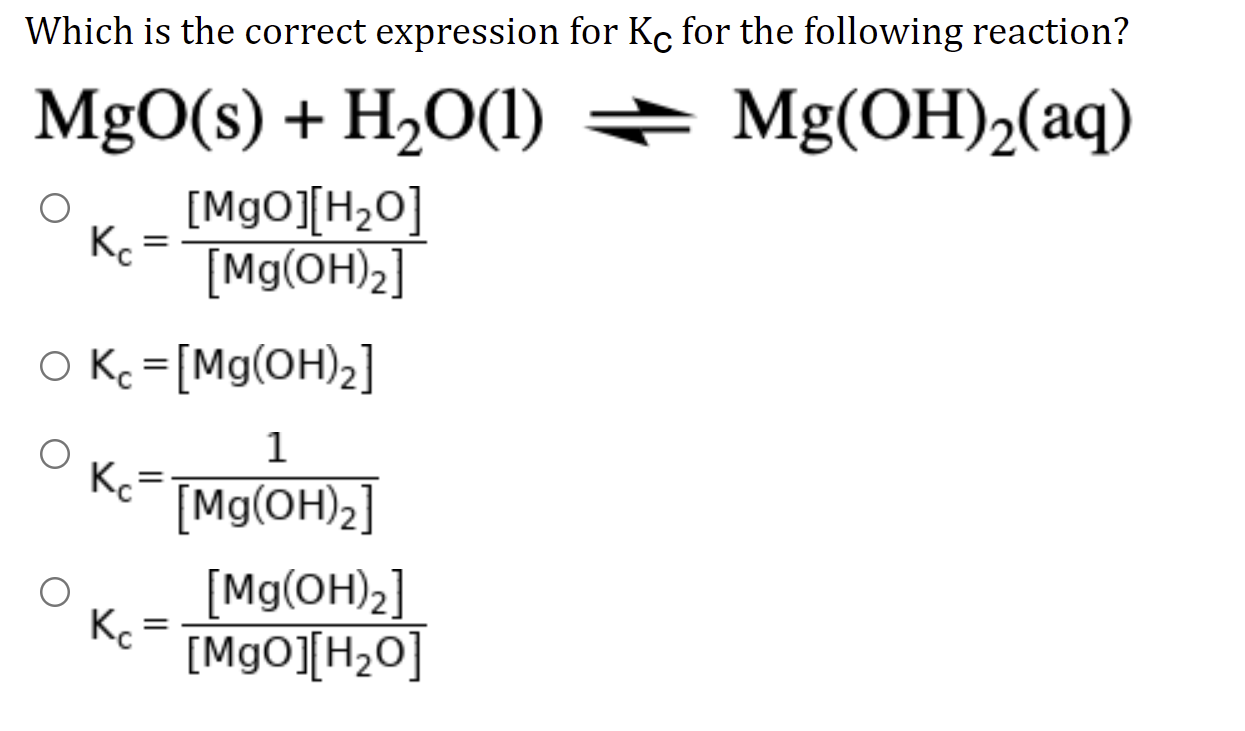Mix formulations of MgO-H2O (L-Asp) samples (wt. % of the solid) and... | Download Scientific Diagram

The transport properties of the MgO–H2O compounds (a-b) a, b Green,... | Download Scientific Diagram
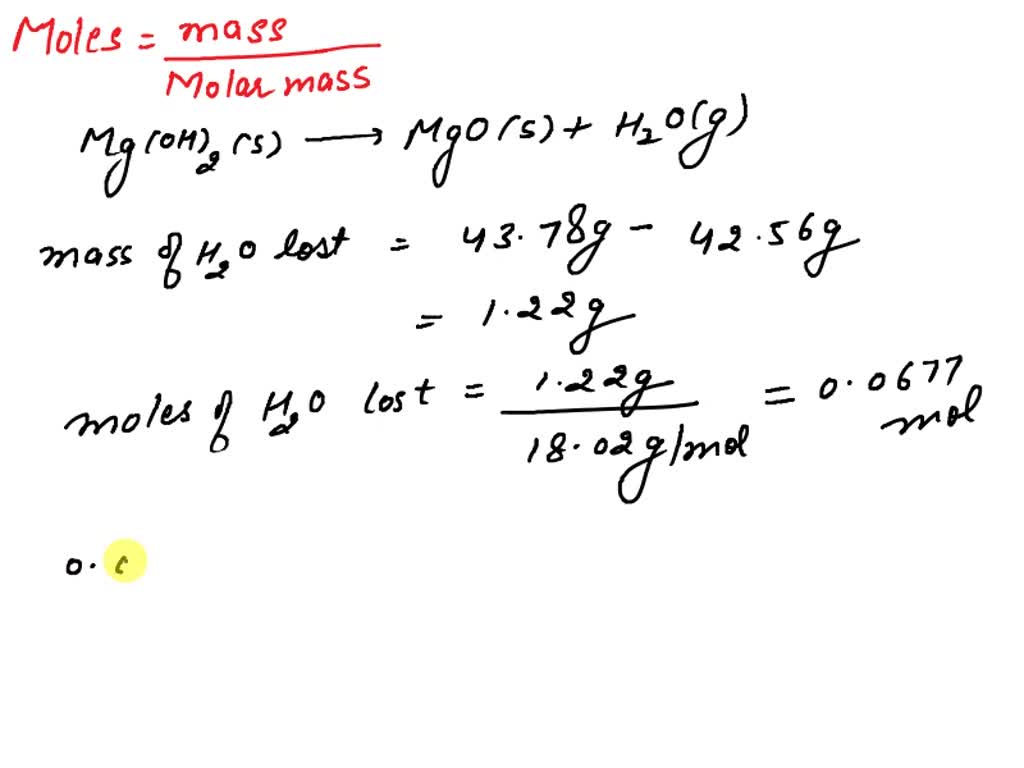
SOLVED: Mg(OH)2(s) = MgO + H2O(g). A sample of Mg(OH)2 was heated in a crucible, the weight of the crucible and its contents went from 43.78g to 42.56g. How many grams of
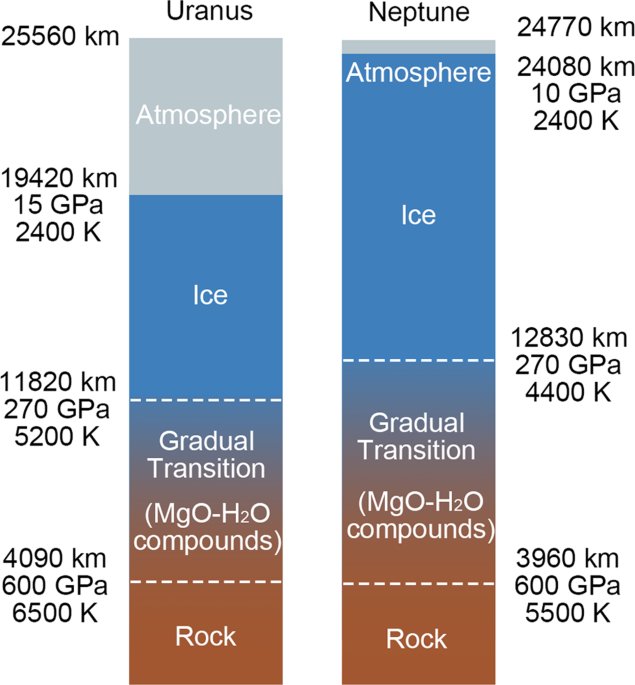
Magnesium oxide-water compounds at megabar pressure and implications on planetary interiors | Nature Communications

The absolute enthalpy of neutralisation of the reaction : MgO(s) +2HCl(aq) → MgCl2(aq)+H2O(1) will be: - 57.33 kJ mol- 2. -57.33 kJ mol-1 Greater than-57.33 kJ moll Less than 57.33 kJ mol-

Mix formulations of MgO-H2O (L-Asp) samples (wt. % of the solid) and... | Download Scientific Diagram

Formation enthalpy and crystal structure of MgO–H2O compounds a Convex... | Download Scientific Diagram

Uzupełnij reakcje wodorotlenku. MgO + H2O --------> ??? LiO + H2O -------> ??? FeO + H2O -------> - Brainly.pl