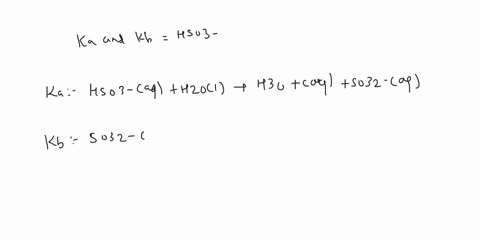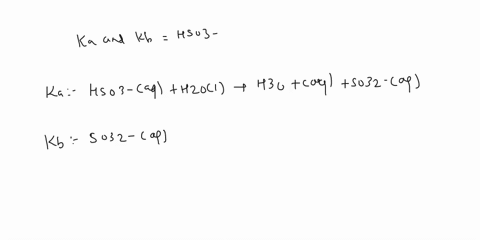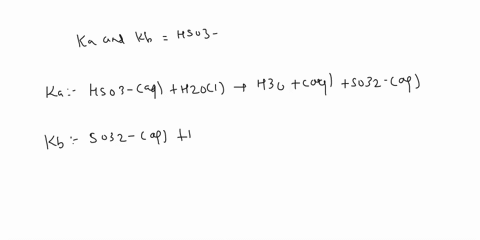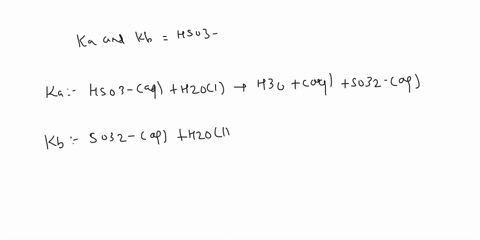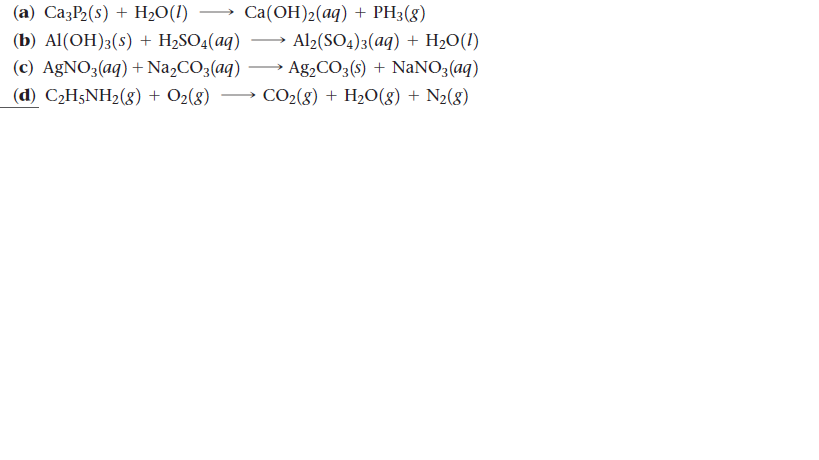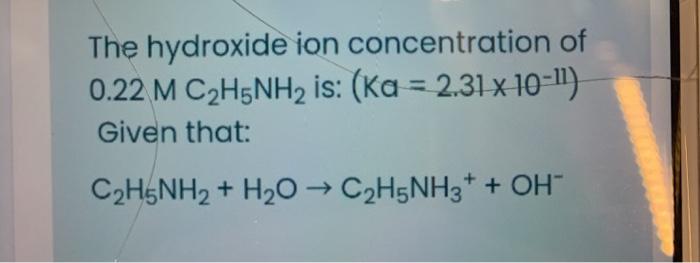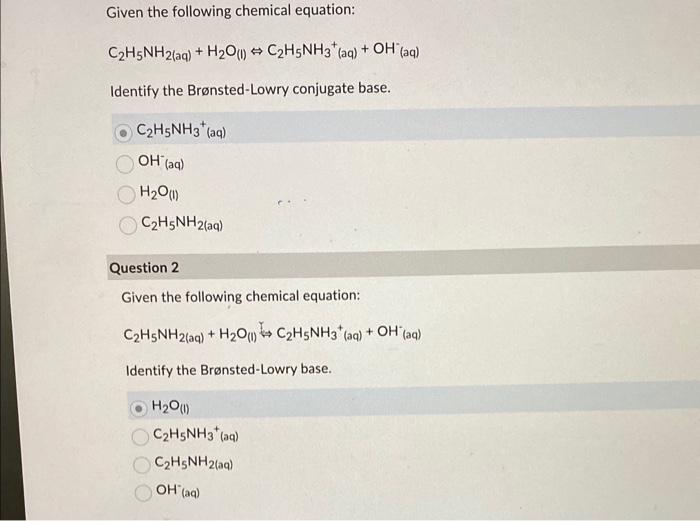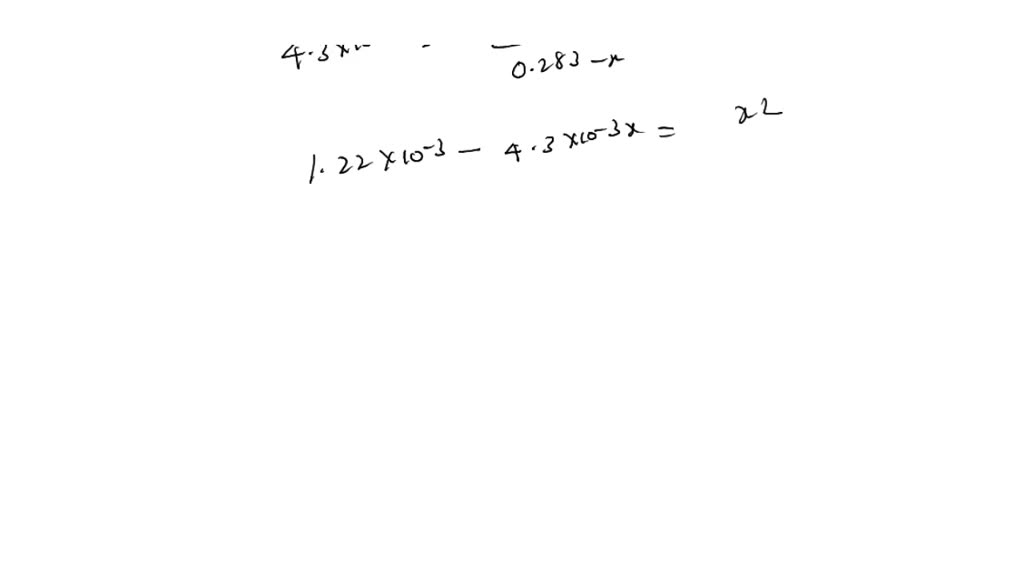
SOLVED: C2H5NH2 + H2O <–> C2H5NH3 +OH- A 0.283 M solution of C2H3NH2 was created. Calculate the equilibrium concentrations of all species, and the pH of the solution.
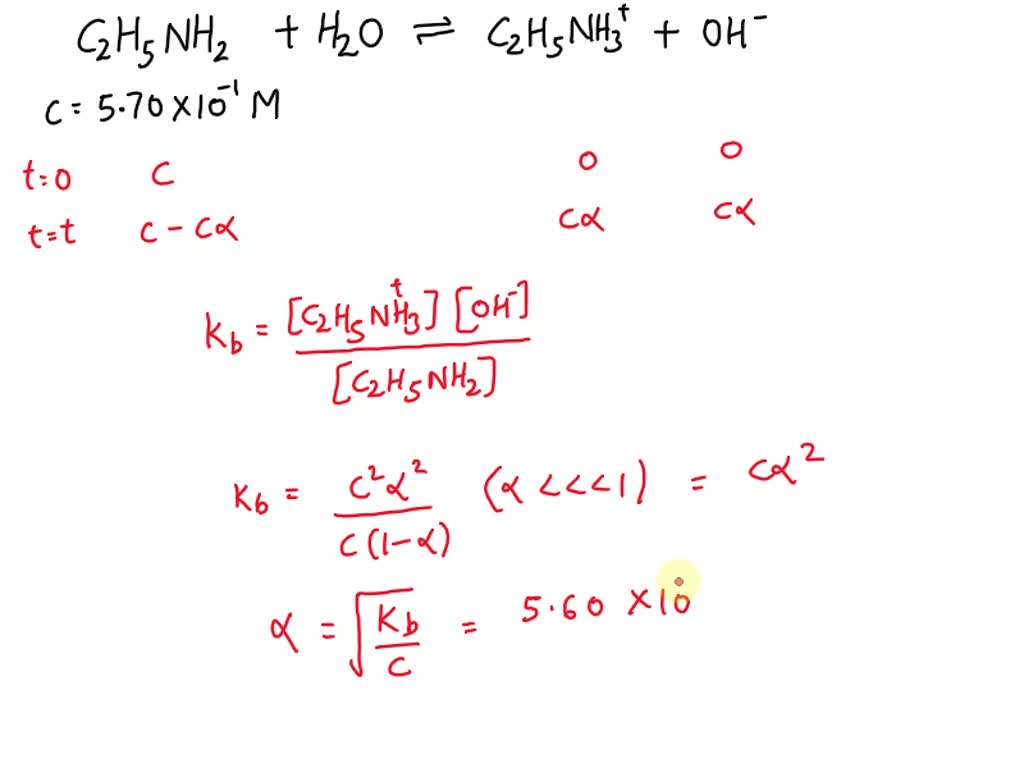
SOLVED: Calculate the pH of a 5.70×10^-1 M aqueous solution of ethylamine hydrochloride (C2H5NH3Cl). (For ethylamine, C2H5NH2, Kb = 5.60×10^-4.)
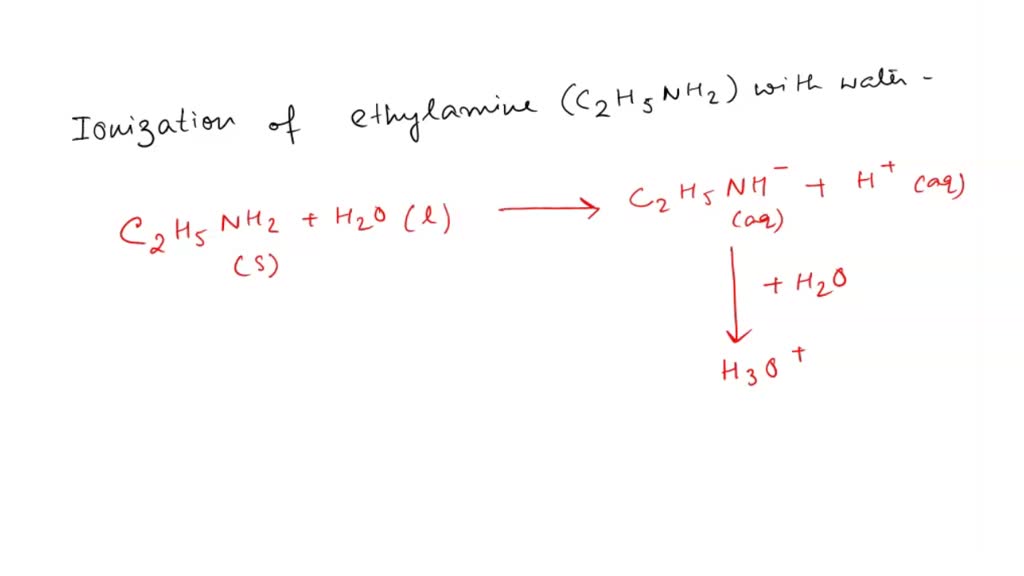
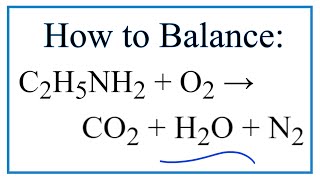
SOLVED: Write the equation for the ionization of ethylamine (C2H5NH2), a weak molecular base, with water