
85. For the reaction, 2NO + 2H2 — N2 + 2H20, the mechanism is given below 2NO=N2O2 N2O2 + H2 - slow > N2O + H2O 4 ) N2O + H2 -
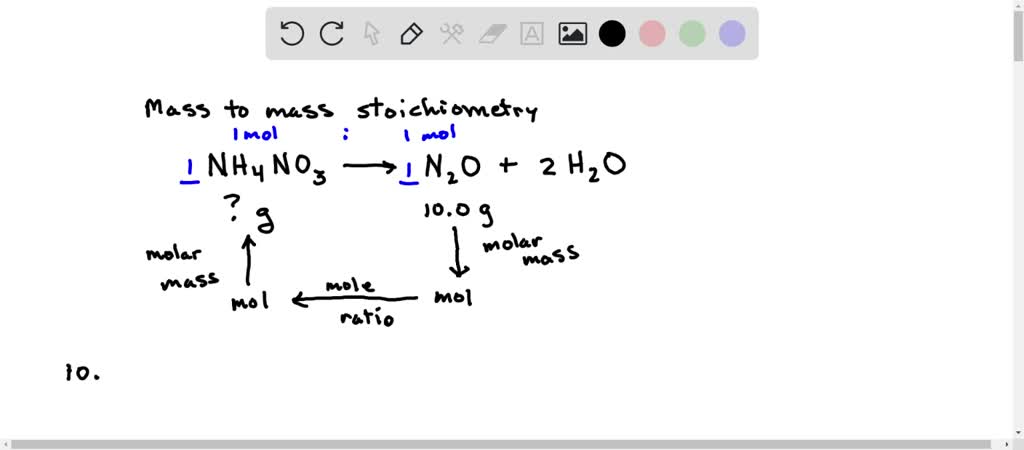
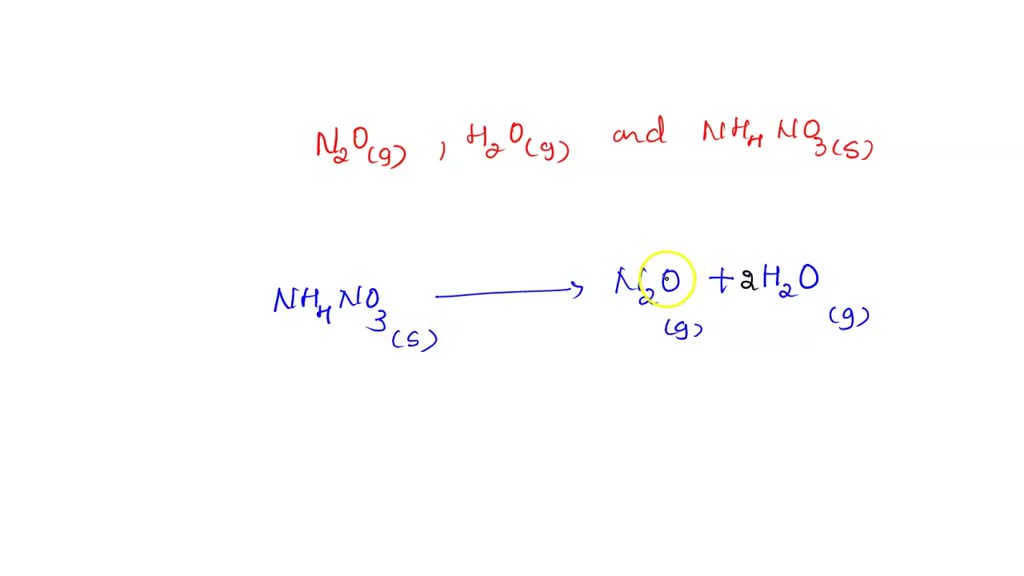
SOLVED: Considering the following balanced equation. NH4NO3 (s) → N2O (g) + 2 H2O (g) Calculate the mass (g) of NH4NO3 needed to produce 10.0 g of nitrous oxide. The molar masses

The unimportance of the reaction H2 + N2O ⇆ H2O + N2: A shock-tube study using H2O time histories and ignition delay times - ScienceDirect

The Isotopocule values (SPN2O and δ¹⁸ON2O/H2O) of soil‐borne N2O from... | Download Scientific Diagram

Simultaneous Monitoring of Atmospheric CH4, N2O, and H2O Using a Single Gas Sensor Based on Mid-IR Quartz-Enhanced Photoacoustic Spectroscopy | Analytical Chemistry

Figure 1 from Structure and Abundance of Nitrous Oxide Complexes in Earth's Atmosphere. | Semantic Scholar
The molar heat of formation of NH4NO3 (s) is -367.57 kJ and those of N2O(g), H2O(l) are 81.46 - Sarthaks eConnect | Largest Online Education Community
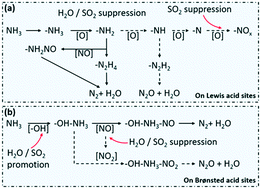
Suppression of N2O formation by H2O and SO2 in the selective catalytic reduction of NO with NH3 over a Mn/Ti–Si catalyst - Catalysis Science & Technology (RSC Publishing)









![Gujrati] Balance the equations Mg + NO3^(-) to Mg^(2+) + N2O + H2O) Gujrati] Balance the equations Mg + NO3^(-) to Mg^(2+) + N2O + H2O)](https://static.doubtnut.com/ss/web-overlay-thumb/5987293.webp)


