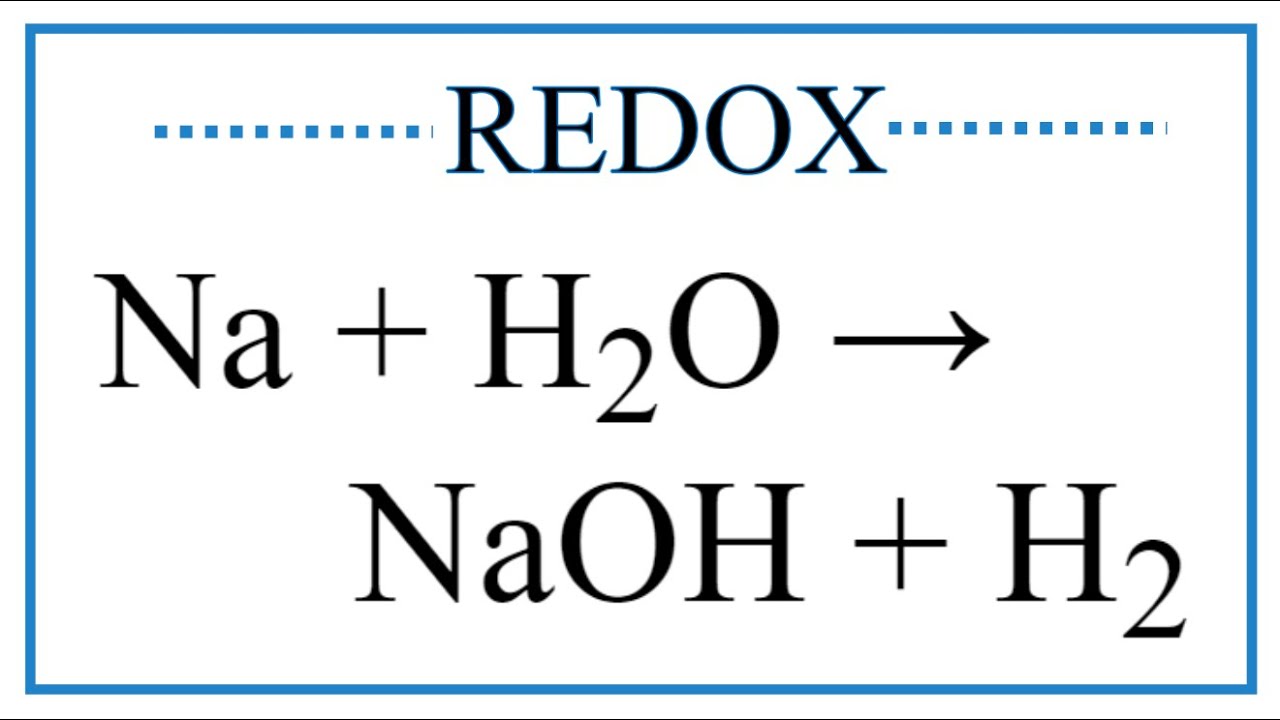
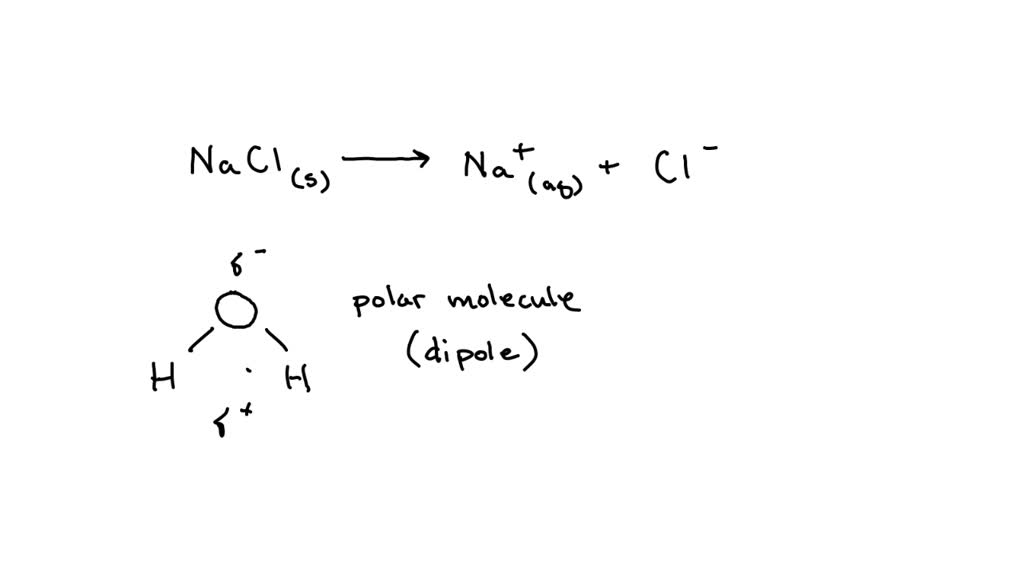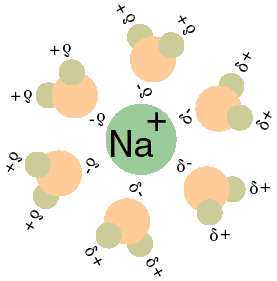![Frontiers | Hydrated Sodium Ion Clusters [Na+(H2O)n (n = 1–6)]: An ab initio Study on Structures and Non-covalent Interaction Frontiers | Hydrated Sodium Ion Clusters [Na+(H2O)n (n = 1–6)]: An ab initio Study on Structures and Non-covalent Interaction](https://www.frontiersin.org/files/Articles/468925/fchem-07-00624-HTML/image_m/fchem-07-00624-g001.jpg)
Frontiers | Hydrated Sodium Ion Clusters [Na+(H2O)n (n = 1–6)]: An ab initio Study on Structures and Non-covalent Interaction

Structures of (a) H2O, (b) Mg(H2O)62+, and (c) Fe(H2O)63+ and (d) the... | Download Scientific Diagram
![Frontiers | Hydrated Sodium Ion Clusters [Na+(H2O)n (n = 1–6)]: An ab initio Study on Structures and Non-covalent Interaction Frontiers | Hydrated Sodium Ion Clusters [Na+(H2O)n (n = 1–6)]: An ab initio Study on Structures and Non-covalent Interaction](https://www.frontiersin.org/files/Articles/468925/fchem-07-00624-HTML/image_m/fchem-07-00624-t002.jpg)
Frontiers | Hydrated Sodium Ion Clusters [Na+(H2O)n (n = 1–6)]: An ab initio Study on Structures and Non-covalent Interaction

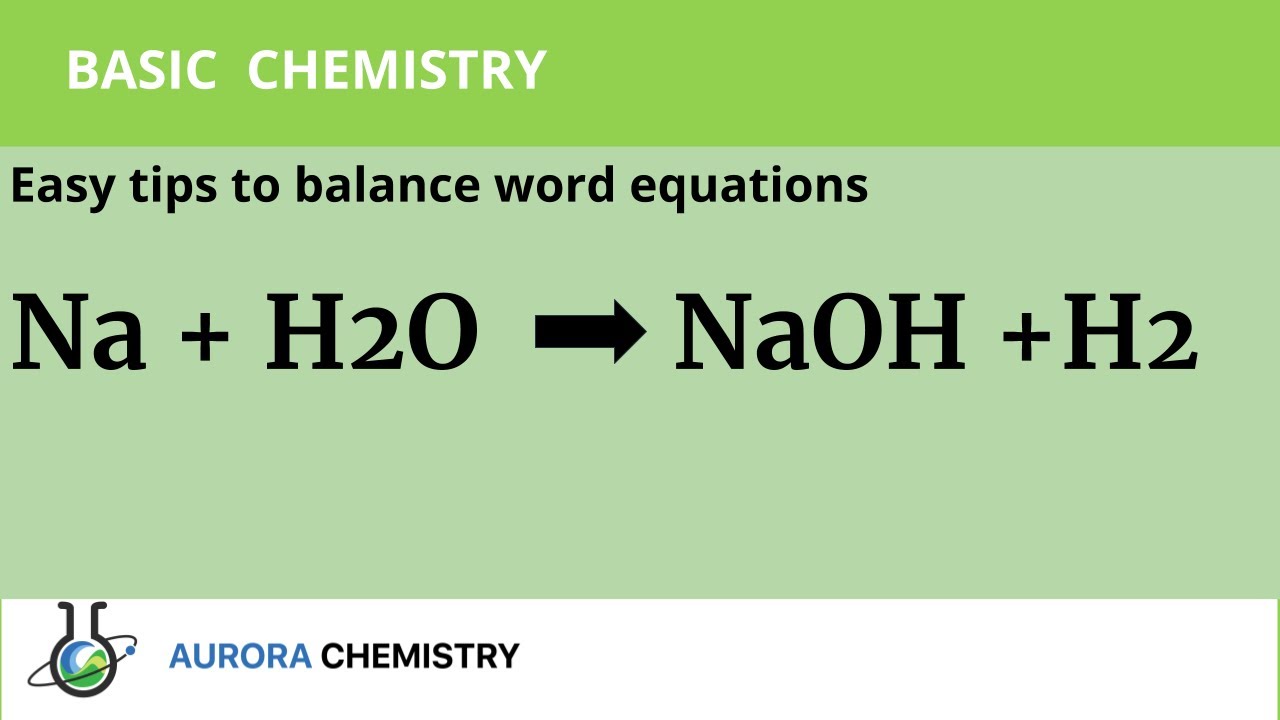
Study of the solubility, viscosity and density in Na+, Zn2+/Cl− − H2O, Na+ − Zn2+ − (H2PO2)− − H2O, Na+, Cl−/(H2PO2)− − H2O, and Zn2+, Cl−/(H2PO2)− − H2O ternary systems, and in