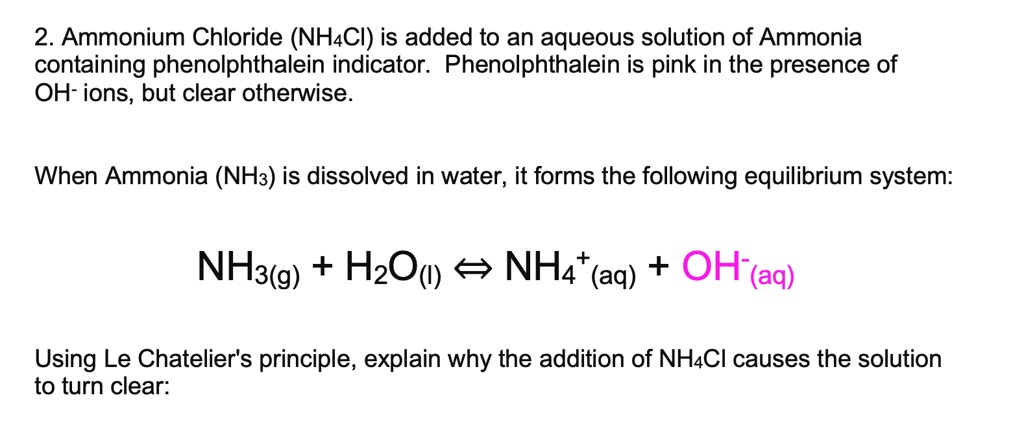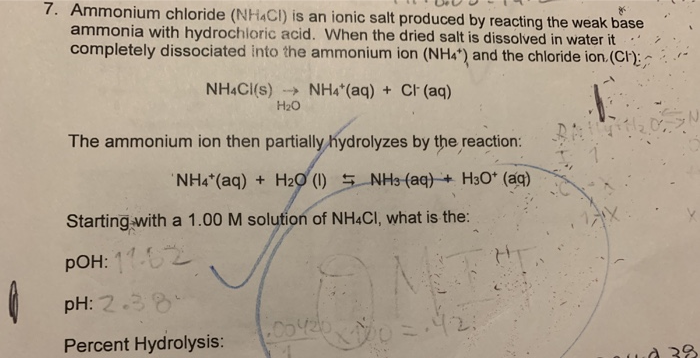![TG and DTG of [Co(EDTA)NH3Cl] H2O (a); TG and DSC of PTh (b) and TG and... | Download Scientific Diagram TG and DTG of [Co(EDTA)NH3Cl] H2O (a); TG and DSC of PTh (b) and TG and... | Download Scientific Diagram](https://www.researchgate.net/profile/M-Dar/publication/316335567/figure/fig2/AS:941601337659404@1601506661415/XRD-pattern-of-CoEDTANH3Cl-H2O-a-and-PTh-CoEDTANH3Cl-H2O-nanocomposite-b_Q320.jpg)
TG and DTG of [Co(EDTA)NH3Cl] H2O (a); TG and DSC of PTh (b) and TG and... | Download Scientific Diagram

The equation for dissolving crystals of ammonium chloride in water at SLC is: NH4Cl(s) + H2O NH4+(aq) + Cl (aq); DH = 15.15 kJ/mol. Is this reaction endothermic or exothermic? | Homework.Study.com

The dissolution of ammonium chloride in water is an endothermic process but still it dissolves in - YouTube

For a ammonium chloride solution in water, the given equilibrium reaction occur:NH 4+ aq + H 2 O l hydrolysis ⇌ NH 4 OH aq + H +aqWhich of the following describes

SOLVED: Write the net ionic equation for the acid-base hydrolysis equilibrium that is established when ammonium chloride is dissolved in water. (Use H3O+ instead of H+.) NH4+(aq) + H2O(l) = NH3(aq) +
Equilibrium phase diagram for ammonium chloride-water solution. [25] We... | Download Scientific Diagram
![TG and DTG of [Co(EDTA)NH3Cl] H2O (a); TG and DSC of PTh (b) and TG and... | Download Scientific Diagram TG and DTG of [Co(EDTA)NH3Cl] H2O (a); TG and DSC of PTh (b) and TG and... | Download Scientific Diagram](https://www.researchgate.net/profile/M-Dar/publication/316335567/figure/fig1/AS:941601337667594@1601506661354/FTIR-spectra-of-CoEDTANH3Cl-H2O-a-PTh-b-and-PTh-CoEDTANH3Cl-H2O_Q320.jpg)
TG and DTG of [Co(EDTA)NH3Cl] H2O (a); TG and DSC of PTh (b) and TG and... | Download Scientific Diagram